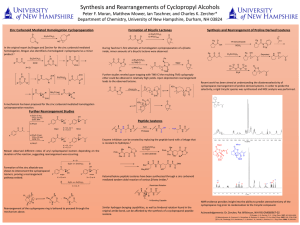
Organic molecules with functional groups containing oxygen
... fertilizer (natural gas (CH4) is one of the raw materials used in its manufacture), energy used (machinery/transport/ ...
... fertilizer (natural gas (CH4) is one of the raw materials used in its manufacture), energy used (machinery/transport/ ...
Enantioselective Henry Reactions under Dual Lewis Acid/Amine
... Claudio Palomo,* Mikel Oiarbide, and Antonio Laso There is increasing interest in developing catalytic asymmetric C C bond-forming processes.[1] In this endeavor the Henry reaction[2] is prominent because of the versatile chemistry of the nitro group.[3] Remarkably, however, while this reaction is c ...
... Claudio Palomo,* Mikel Oiarbide, and Antonio Laso There is increasing interest in developing catalytic asymmetric C C bond-forming processes.[1] In this endeavor the Henry reaction[2] is prominent because of the versatile chemistry of the nitro group.[3] Remarkably, however, while this reaction is c ...
Wed March 3 lecture
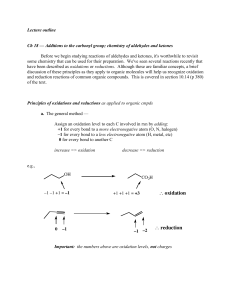
... Before we begin studying reactions of aldehydes and ketones, it's worthwhile to revisit some chemistry that can be used for their preparation. We've seen several reactions recently that have been described as oxidations or reductions. Although these are familiar concepts, a brief discussion of these ...
... Before we begin studying reactions of aldehydes and ketones, it's worthwhile to revisit some chemistry that can be used for their preparation. We've seen several reactions recently that have been described as oxidations or reductions. Although these are familiar concepts, a brief discussion of these ...
Chapter 3. The Concept of Protecting Functional Groups
... It is readily introduced by reaction of the enol ether dihyrdopyran with an alcohol in the presence of an acid catalyst such as TsOH, BF3, POCl3. THP group is readily hydrolyzed under aqueous acidic conditions with AcOH-THF, TsOH, Dowex-H (cation exchange resin) ...
... It is readily introduced by reaction of the enol ether dihyrdopyran with an alcohol in the presence of an acid catalyst such as TsOH, BF3, POCl3. THP group is readily hydrolyzed under aqueous acidic conditions with AcOH-THF, TsOH, Dowex-H (cation exchange resin) ...
Aldehydes and Ketones
... carbonyl carbon, but the “1” is usually omitted from the name. The ring is then numbered clockwise or counterclockwise to give the first substituent the lower number. ...
... carbonyl carbon, but the “1” is usually omitted from the name. The ring is then numbered clockwise or counterclockwise to give the first substituent the lower number. ...
n - TU Chemnitz
... a) S. Kirchmeyer, A. Mertens, G. A. Olah, Synthesis 1983, 500–502; b) A. Hassner, R. Fibiger, A. S. Amarasekara, J. Org. Chem. 1988, 53, 22–27. a) P. Herczegh, M. Zsély, I. Kovács, G. Batta, F. J. Sztaricskai, Tetrahedron Lett. 1990, 31, 1195–1198. b) C. Gauthier, Y. Ramondenc, G. Plé, ...
... a) S. Kirchmeyer, A. Mertens, G. A. Olah, Synthesis 1983, 500–502; b) A. Hassner, R. Fibiger, A. S. Amarasekara, J. Org. Chem. 1988, 53, 22–27. a) P. Herczegh, M. Zsély, I. Kovács, G. Batta, F. J. Sztaricskai, Tetrahedron Lett. 1990, 31, 1195–1198. b) C. Gauthier, Y. Ramondenc, G. Plé, ...
Study Guide for Exam 2-‐ Aldehydes and Ketones
... E X A M P R E P A R A T I O N Nomenclature: Aldehydes and Ketones Syntheses: This is the first exam with targeted syntheses that you need to work out prior to the exam. You are free ...
... E X A M P R E P A R A T I O N Nomenclature: Aldehydes and Ketones Syntheses: This is the first exam with targeted syntheses that you need to work out prior to the exam. You are free ...
Lecture 14a - UCLA Chemistry and Biochemistry
... R= alkyl (Me, Et, iso-Pr, tert. Bu), alkoxy (OMe, OEt), amino ...
... R= alkyl (Me, Et, iso-Pr, tert. Bu), alkoxy (OMe, OEt), amino ...
ALDEHYDES , KETONES AND CARBOXYLIC ACIDS
... Clemmensen reduction: The carbonyl group of Wolff-Kishner reduction: The carbonyl group of aldehydes and ketones is reduced to CH2 group on aldehydes and ketones is reduced to CH2 group on treatment with zinc-amalgam and concentrated treatment with hydrazine followed by heating with ...
... Clemmensen reduction: The carbonyl group of Wolff-Kishner reduction: The carbonyl group of aldehydes and ketones is reduced to CH2 group on aldehydes and ketones is reduced to CH2 group on treatment with zinc-amalgam and concentrated treatment with hydrazine followed by heating with ...
aldehydes and ketones
... • Using the root alkane name, drop the –e ending and change to –one. • Number the longest carbon chain so the C=O group has the lowest number. • Name and number other substituents as before. • Examples: ...
... • Using the root alkane name, drop the –e ending and change to –one. • Number the longest carbon chain so the C=O group has the lowest number. • Name and number other substituents as before. • Examples: ...
Aldol reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry.Discovered independently by Charles-Adolphe Wurtz and Alexander Borodin in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as aldols, from the aldehyde + alcohol, a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic.For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritoland the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt).The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the α- and β-carbon of the aldol adduct, marked with asterisks in the scheme below) are formed. Modern methodology is capable of not only allowing aldol reactions to proceed in high yield but also controlling both the relative and absolute stereochemical configuration of these stereocenters. This ability to selectively synthesize a particular stereoisomer is significant because different stereoisomers can have very different chemical and biological properties.For example, stereogenic aldol units are especially common in polyketides, a class of molecules found in biological organisms. In nature, polyketides are synthesized by enzymes that effect iterative Claisen condensations. The 1,3-dicarbonyl products of these reactions can then be variously derivatized to produce a wide variety of interesting structures. Often, such derivitization involves the reduction of one of the carbonyl groups, producing the aldol subunit. Some of these structures have potent biological properties: the immunosuppressant FK506, the anti-tumor agent discodermolide, or the antifungal agent amphotericin B, for example. Although the synthesis of many such compounds was once considered nearly impossible, aldol methodology has allowed their efficient synthesis in many cases.A typical modern aldol addition reaction, shown above, might involve the nucleophilic addition of a ketone enolate to an aldehyde. Once formed, the aldol product can sometimes lose a molecule of water to form an α,β-unsaturated carbonyl compound. This is called aldol condensation. A variety of nucleophiles may be employed in the aldol reaction, including the enols, enolates, and enol ethers of ketones, aldehydes, and many other carbonyl compounds. The electrophilic partner is usually an aldehyde or ketone (many variations, such as the Mannich reaction, exist). When the nucleophile and electrophile are different, the reaction is called a crossed aldol reaction; on the converse, when the nucleophile and electrophile are the same, the reaction is called an aldol dimerization.