Class Notes
... Concept 5.1 Most macromolecules are polymers, built from monomers. Three of the four classes of macromolecules—carbohydrates, proteins, and nucleic acids—form chain-like molecules called polymers. ○ A polymer is a long molecule consisting of many similar or identical building blocks linked by cova ...
... Concept 5.1 Most macromolecules are polymers, built from monomers. Three of the four classes of macromolecules—carbohydrates, proteins, and nucleic acids—form chain-like molecules called polymers. ○ A polymer is a long molecule consisting of many similar or identical building blocks linked by cova ...
STRUCTURAL ORGANIZATION OF LIVING SYSTEMS At all levels
... hydration shell around protein molecules is 0.4 - 0.5 nm thick, and its mass can reach as much as 30% of that of the mass of the protein. In the B-helix structure of DNA 10 water molecules bind to every basepair. ...
... hydration shell around protein molecules is 0.4 - 0.5 nm thick, and its mass can reach as much as 30% of that of the mass of the protein. In the B-helix structure of DNA 10 water molecules bind to every basepair. ...
Example: search for regulatory binding sites
... y, w, in a protein. Because w is around 180O, the backbone configuration can be specified by f and y, for each peptide unit. ...
... y, w, in a protein. Because w is around 180O, the backbone configuration can be specified by f and y, for each peptide unit. ...
3.the nature of proteins
... Myoglobin is a globular protein displaying the tertiary level of structure Myoglobin is a protein found in muscle cells Proteins consisting of more than one polypeptide chain display quaternary structure Haemoglobin is a protein consisting of more than one polypeptide chain Haemoglobin co ...
... Myoglobin is a globular protein displaying the tertiary level of structure Myoglobin is a protein found in muscle cells Proteins consisting of more than one polypeptide chain display quaternary structure Haemoglobin is a protein consisting of more than one polypeptide chain Haemoglobin co ...
Question Paper
... ii) Write thermo chemical equation for vaporisation of Ethanol (C2H5OH). iii) Calculate the enthalpy of vapourisation of Ethanol, given enthalpies of formation of liquid Ethanol and gaseous Ethanol as – 277.6 kJ and -235.4 kJ respectively. 22. a) ...
... ii) Write thermo chemical equation for vaporisation of Ethanol (C2H5OH). iii) Calculate the enthalpy of vapourisation of Ethanol, given enthalpies of formation of liquid Ethanol and gaseous Ethanol as – 277.6 kJ and -235.4 kJ respectively. 22. a) ...
Active Site Interactions in Oligomeric Structures of Inorganic
... The consequences of single amino acid substitutions at the active site become clear from the comparison of Asp-97Asn and Asp-97Glu substitutions in EPPases and Asp-147Glu replacement in Y-PPase. The properties of these mutant forms are well studied: the dependences of kcat on pH and dissociation ...
... The consequences of single amino acid substitutions at the active site become clear from the comparison of Asp-97Asn and Asp-97Glu substitutions in EPPases and Asp-147Glu replacement in Y-PPase. The properties of these mutant forms are well studied: the dependences of kcat on pH and dissociation ...
Lorem Ipsum - Tri-County Technical College
... group is removed from amino acids The result is a keto acid Keto acids enter the respiratory cycle as pyruvic acid or as one of the other types of molecules found in the Kreb’s cycle. The amino group is converted to ammonia ...
... group is removed from amino acids The result is a keto acid Keto acids enter the respiratory cycle as pyruvic acid or as one of the other types of molecules found in the Kreb’s cycle. The amino group is converted to ammonia ...
Carbon Compounds 2-3 Foldable Instructions
... On a food label, there is a total # of carbs. Below that, it lists the # of grams of dietary fiber (if this is missing, there is no fiber in that food) and # of grams of sugar. Starches are not listed on food labels. To figure out the amount of starch in a food, subtract fiber and sugar from the t ...
... On a food label, there is a total # of carbs. Below that, it lists the # of grams of dietary fiber (if this is missing, there is no fiber in that food) and # of grams of sugar. Starches are not listed on food labels. To figure out the amount of starch in a food, subtract fiber and sugar from the t ...
Cellular Respiration NOTES
... Cellular respiration converts the energy stored in the bonds of the glucose into energy in ATP. Why is it important? – all living organisms need to convert the energy in the food they eat (or in the case of plants in the food they produce through photosynthesis) into a form of energy that is easy to ...
... Cellular respiration converts the energy stored in the bonds of the glucose into energy in ATP. Why is it important? – all living organisms need to convert the energy in the food they eat (or in the case of plants in the food they produce through photosynthesis) into a form of energy that is easy to ...
WHAT THEY DO
... Enzymes speed up chemical reactions (catalysts) They are required for all biochemical reactions Stay tuned…We will learn more about enzymes later in this week! ...
... Enzymes speed up chemical reactions (catalysts) They are required for all biochemical reactions Stay tuned…We will learn more about enzymes later in this week! ...
Presentation
... • Inhibitor binds only to free enzyme (E) not (ES) • Substrate cannot bind when I is bound at active site (S and I “compete” for the enzyme active site) ...
... • Inhibitor binds only to free enzyme (E) not (ES) • Substrate cannot bind when I is bound at active site (S and I “compete” for the enzyme active site) ...
File
... A molecule is oxidized if it loses an electron or hydrogen atom and is reduced when it gains an electron or hydrogen atom. Collectively, the two processes are referred to as a redox reaction because when one molecule is oxidized, another is reduced. ...
... A molecule is oxidized if it loses an electron or hydrogen atom and is reduced when it gains an electron or hydrogen atom. Collectively, the two processes are referred to as a redox reaction because when one molecule is oxidized, another is reduced. ...
Electronic Spectra
... Charge transfer, either ligand to metal or metal to ligand. These are often extremely intense and are generally found in the UV but they may have a tail into the visible. d-d, these can occur in both the UV and visible region but since they are forbidden transitions have small intensities. Re ...
... Charge transfer, either ligand to metal or metal to ligand. These are often extremely intense and are generally found in the UV but they may have a tail into the visible. d-d, these can occur in both the UV and visible region but since they are forbidden transitions have small intensities. Re ...
Chapter 1 OBJECTIVES
... controlled reaction. • The higher the substrate concentration, the faster the reaction - up to a limit. • If substrate concentration is high enough, the enzyme becomes saturated with substrate. (The active sites of all enzymes molecules are engaged.) • When an enzyme is saturated, the reaction rate ...
... controlled reaction. • The higher the substrate concentration, the faster the reaction - up to a limit. • If substrate concentration is high enough, the enzyme becomes saturated with substrate. (The active sites of all enzymes molecules are engaged.) • When an enzyme is saturated, the reaction rate ...
Lecture 10 Protein Tertiary (3D) Structure
... • Database of molecular structures – Obtained by crystallography or NMR – Carefully curated and validated ...
... • Database of molecular structures – Obtained by crystallography or NMR – Carefully curated and validated ...
Class11 POGIL Translation Full Win17 all pages
... b. How many tRNAs will bind to the ribosome to make this protein? _________ c. Which of the following sequences within the mRNA most likely contains the ribosome binding site? (Circle ONE) 5'UAGCUAGCA3' 5'UUAAUGG3' 5'AAGGAGGC3' ...
... b. How many tRNAs will bind to the ribosome to make this protein? _________ c. Which of the following sequences within the mRNA most likely contains the ribosome binding site? (Circle ONE) 5'UAGCUAGCA3' 5'UUAAUGG3' 5'AAGGAGGC3' ...
PREVIEW_on_Ng_etal_STRUCTURE-MK
... et al, 2013). More recently, a fifth family was identified in diarrhea-causing strains of Escherichia coli and Citrobacter freundii represented by toxins EcxAB and CfxAB, respectively (Jansson et al, 2010; Karasawa et al, 2002). In the present issue of Structure, Ng, Littler and co-workers present t ...
... et al, 2013). More recently, a fifth family was identified in diarrhea-causing strains of Escherichia coli and Citrobacter freundii represented by toxins EcxAB and CfxAB, respectively (Jansson et al, 2010; Karasawa et al, 2002). In the present issue of Structure, Ng, Littler and co-workers present t ...
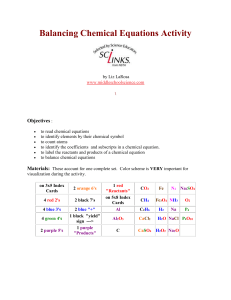
Balancing Chemical Equations Activity by Liz LaRosa www
... The index cards are a bit time consuming to create. I had some students help at lunch time for a few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making in ...
... The index cards are a bit time consuming to create. I had some students help at lunch time for a few days. Once done, you can laminate them and have them forever! The materials account for one complete set which is good for 2-3 students to use. Print activity cards on card stock instead of making in ...
How to Make a Collage
... 19. An organic compound which has the empirical formula CHO has a molar mass of 232. Its molecular formula is: a) ...
... 19. An organic compound which has the empirical formula CHO has a molar mass of 232. Its molecular formula is: a) ...
Document
... carbon, hydrogen, and oxygen? a. sugar b. water c. salt d. sulfuric acid _____ 2. Which sof the following is NOT true about electrons when chemical bonds form? a. Electrons are shared. b. Electrons are lost. c. Electrons are destroyed d. Electrons are gained. _____ 3. Which of the following is an in ...
... carbon, hydrogen, and oxygen? a. sugar b. water c. salt d. sulfuric acid _____ 2. Which sof the following is NOT true about electrons when chemical bonds form? a. Electrons are shared. b. Electrons are lost. c. Electrons are destroyed d. Electrons are gained. _____ 3. Which of the following is an in ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.