Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Analyze: We are given several chemical formulas and asked to classify each substance as a strong electrolyte, weak electrolyte, or nonelectrolyte. Plan: The approach we take is outlined in Table 4.3. We can predict whether a substance is ionic or molecular, based on its composition. As we saw in Sec ...
... Analyze: We are given several chemical formulas and asked to classify each substance as a strong electrolyte, weak electrolyte, or nonelectrolyte. Plan: The approach we take is outlined in Table 4.3. We can predict whether a substance is ionic or molecular, based on its composition. As we saw in Sec ...
Handout-5
... include octahedral and square-planar complexes. The rule is not applicable to tetrahedral complexes as it does not contain a center of symmetry. ...
... include octahedral and square-planar complexes. The rule is not applicable to tetrahedral complexes as it does not contain a center of symmetry. ...
6.8-6.10 Citric acid cycle and Oxidative phosphorylation
... • Pyruvate does not enter the citric acid cycle, but undergoes some chemical grooming in which – a carboxyl group is removed and given off as CO2, – the two-carbon compound remaining is oxidized while a molecule of NAD+ is reduced to NADH, – coenzyme A joins with the two-carbon group to form acetyl ...
... • Pyruvate does not enter the citric acid cycle, but undergoes some chemical grooming in which – a carboxyl group is removed and given off as CO2, – the two-carbon compound remaining is oxidized while a molecule of NAD+ is reduced to NADH, – coenzyme A joins with the two-carbon group to form acetyl ...
Final a
... assuming the production of one glucose molecule from carbon dioxide. How many turnovers are required from Rubisco for each glucose produced? ...
... assuming the production of one glucose molecule from carbon dioxide. How many turnovers are required from Rubisco for each glucose produced? ...
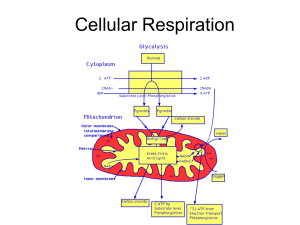
Cellular Respiration
... • Glycolysis = 2 ATP (actually anaerobic) • Citric Acid Cycle = 2 ATP • Electron Transport Chain = 32 ATP • 1 Glucose = 36 TOTAL ATP for aerobic respiration ...
... • Glycolysis = 2 ATP (actually anaerobic) • Citric Acid Cycle = 2 ATP • Electron Transport Chain = 32 ATP • 1 Glucose = 36 TOTAL ATP for aerobic respiration ...
Protein design TOPS cartoon of Top7 Approach to designing Top7
... (iii) Optimise backbone structure optimise φ and ψ again in a 10-residue window around the per turbation site. Measure energy again, and use Metropolis criterion to decide whether to accept or reject. Steps (i), (ii) and (iii) are repeated several thousand times. After every 20 such moves, a full co ...
... (iii) Optimise backbone structure optimise φ and ψ again in a 10-residue window around the per turbation site. Measure energy again, and use Metropolis criterion to decide whether to accept or reject. Steps (i), (ii) and (iii) are repeated several thousand times. After every 20 such moves, a full co ...
Chapter 2 ppt B
... • pH change interferes with cell function and may damage living tissue • Even slight change in pH can be fatal • pH is regulated by kidneys, lungs, and chemical buffers ...
... • pH change interferes with cell function and may damage living tissue • Even slight change in pH can be fatal • pH is regulated by kidneys, lungs, and chemical buffers ...
in Graminaceous Plants
... Nicotianamine aminotransferase (NAAT), the key enzyme involved in the biosynthesis of mugineic acid family phytosiderophores (MAs), catalyzes the amino transfer of nicotianamine (NA). MAs are found only in graminaceous plants, although NA has been detected in every plant so far investigated. Therefo ...
... Nicotianamine aminotransferase (NAAT), the key enzyme involved in the biosynthesis of mugineic acid family phytosiderophores (MAs), catalyzes the amino transfer of nicotianamine (NA). MAs are found only in graminaceous plants, although NA has been detected in every plant so far investigated. Therefo ...
unit 4: chemical reaction rates
... US specific outcomes The following specific outcomes are covered in this module: ...
... US specific outcomes The following specific outcomes are covered in this module: ...
Metabolism of amino acids
... Amino nitrogen released from carbon sceletons of AAs can be transported in blood as a) NH4+ physiologically up to 35 µmol/l (NH3 + H + b) alanine ...
... Amino nitrogen released from carbon sceletons of AAs can be transported in blood as a) NH4+ physiologically up to 35 µmol/l (NH3 + H + b) alanine ...
Week of Sept. 20
... Electron Counting Step 1: Determine the oxidation state of the metal. To do this, balance the ligand charges with an equal opposite charge on the metal. This is the metal's formal oxidation state. ...
... Electron Counting Step 1: Determine the oxidation state of the metal. To do this, balance the ligand charges with an equal opposite charge on the metal. This is the metal's formal oxidation state. ...
ACT Science Practice Test 1 ANSWERS File
... b. Experiment 1 establishes that both Beetle A and Beetle B eat whole-wheat flour. c. Experiment 1 establishes the non-competitive total population of each insect. d. Experiment 1 demonstrates that the caterpillar has a much slower growth rate than the beetles. c. Experiment 1 is a control experimen ...
... b. Experiment 1 establishes that both Beetle A and Beetle B eat whole-wheat flour. c. Experiment 1 establishes the non-competitive total population of each insect. d. Experiment 1 demonstrates that the caterpillar has a much slower growth rate than the beetles. c. Experiment 1 is a control experimen ...
Cellular Respiration
... Glycolysis yields 2 molecules of pyruvic acid and each react with coenzyme A to form acetyl CoA. Krebs Cycle- breaks down the acetyl CoA to produce CO2, hydrogen, and ATP. ...
... Glycolysis yields 2 molecules of pyruvic acid and each react with coenzyme A to form acetyl CoA. Krebs Cycle- breaks down the acetyl CoA to produce CO2, hydrogen, and ATP. ...
chemistry 103 - chem.uwec.edu
... Example: Calculate the pH of a buffer system containing 1.0 M CH3CO2H and 1.0 M NaCH3CO2. What is the pH of the buffer after the addition of 0.10 moles of gaseous HCl to 1.00 liter of the buffer solution? The Ka for acetic acid is 1.8 x 10-5. Because acetic acid is a weak acid, we can ignore the sm ...
... Example: Calculate the pH of a buffer system containing 1.0 M CH3CO2H and 1.0 M NaCH3CO2. What is the pH of the buffer after the addition of 0.10 moles of gaseous HCl to 1.00 liter of the buffer solution? The Ka for acetic acid is 1.8 x 10-5. Because acetic acid is a weak acid, we can ignore the sm ...
... Choice A: Briefly describe the molecular basis of the hydrophobic effect and indicate its role in the stability of folded proteins. Choice B: Briefly describe conformational entropy and indicate its role in the stability of folded proteins. Choice C: Briefly describe the role of van der Waals forces ...
Discovery of Enzymes
... The enzyme does not actually form a chemical bond with the substrate. After the reaction, the products are released and the enzyme returns to its normal shape. Because the enzyme does not form chemical bonds with the substrate, it remains unchanged. The enzyme molecule can be reused repeatedly Only ...
... The enzyme does not actually form a chemical bond with the substrate. After the reaction, the products are released and the enzyme returns to its normal shape. Because the enzyme does not form chemical bonds with the substrate, it remains unchanged. The enzyme molecule can be reused repeatedly Only ...
It Works! Shake™ Product Info Sheet
... Yellow Pea and Organic Sprouted Brown Rice proteins. This “perfect protein” blend contains a balanced amino acid profile. What does that mean? These two proteins complement each other, with the Organic Sprouted Brown Rice protein providing a complete spectrum of amino acids, including being high in ...
... Yellow Pea and Organic Sprouted Brown Rice proteins. This “perfect protein” blend contains a balanced amino acid profile. What does that mean? These two proteins complement each other, with the Organic Sprouted Brown Rice protein providing a complete spectrum of amino acids, including being high in ...
Protein thermostability in Archaea and Eubacteria
... However, thermophiles in general have a higher number of disulfide bridges as compared to mesophiles (Mallick et al., 2002). Contrary to this, other findings suggest that disulfide bridges may not be as important for stability in some proteins (Lo et al., 1999; Tanaka et al., 2004). It is possible t ...
... However, thermophiles in general have a higher number of disulfide bridges as compared to mesophiles (Mallick et al., 2002). Contrary to this, other findings suggest that disulfide bridges may not be as important for stability in some proteins (Lo et al., 1999; Tanaka et al., 2004). It is possible t ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.